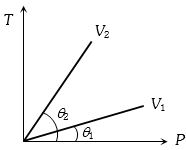
At constant volume, temperature is increased. Then
AIPMT 1989, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1From the following $P-T$ graph what interference can be drawnView Solution
- 2An ideal gas is enclosed in a cylinder at pressure of $2\,atm$ and temperature, $300\,K.$ The mean time between two successive collisions is $6 \times 10^{-8}\, s.$ If the pressure is doubled and temperature is increased to $500\,K,$ the mean time between two successive collisions will be close toView Solution
- 3A sample of gas is at $0°C.$ To what temperature it must be raised in order to double the $r.m.s.$ speed of the molecule ....... $^oC$View Solution
- 4A gas mixture consists of $3\,moles$ of oxygen and $5\,moles$ or argon at temperature $T.$ Considering only translational and rotational modes, the total internal energy of the system isView Solution
- 5Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
- 6The average kinetic energy of a gas molecule at $27^oC$ is $6.21 \times {10^{ - 21}}\,J$. Its average kinetic energy at $227°C$ will beView Solution
- 7If the intermolecular forces vanish away, the volume occupied by the molecules contained in $4.5 \,kg$ water at standard temperature and pressure will beView Solution
- 8View SolutionOn colliding in a closed container the gas molecules
- 9The root mean square velocity of a gas molecule of mass $m$ at a given temperature is proportional toView Solution
- 10View SolutionThe rate of diffusion is
