Consider an ideal gas confined in an isolated closed chamber. As the gas undergoes an adiabatic expansion, the average time of collision between molecules increases as $V ^q$, where $V$ is the volume of the gas. The value of $q$ is $\left( {\gamma = \frac{{{C_P}}}{{{C_V}}}} \right)$
JEE MAIN 2015, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300\ K.$ The ratio of the average rotational kinetic energy per $O_2$ molecule to that per $N_2$ molecule isView Solution
- 2$P-V$ diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will beView Solution
- 3View SolutionAccording to kinetic theory of gases,
- 4A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
- 5View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
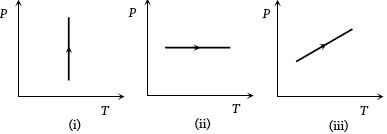
- 6The speeds of $5$ molecules of a gas (in arbitrary units) are as follows : $2, 3, 4, 5, 6.$ The root mean square speed for these molecules isView Solution
- 7According to $C.E.$ van der Waal, the interatomic potential varies with the average interatomic distance $(R)$ asView Solution
- 8An ideal gas equation can be written as $P=\frac{\rho R T}{M_{0}}$ where $\rho$ and $M_{0}$ are respectively,View Solution
- 9The mean kinetic energy of a gas at $300 K$ is $100 J.$ The mean energy of the gas at $450 K$ is equal to ...... $J$View Solution
- 10An $HCl$ molecule has rotational, translational and vibrational motions. If the $rms$ velocity of $HCl$ molecules in its gaseous phase is $\bar v ,\,m$ is its mass and $k_B$ is Boltzmann constant, then its temperature will beView Solution
