An ideal gas equation can be written as $P=\frac{\rho R T}{M_{0}}$ where $\rho$ and $M_{0}$ are respectively,
NEET 2020, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas at $27^o C$ has a volume $V$ and pressure $P.$ On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ...... $^oC$View Solution
- 2$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$View Solution
- 3View SolutionInternal forces acting in a system of particle can change
- 4A vessel contains $1$ mole of ${O_2}$ gas (molar mass $32)$ at a temperature $T.$ The pressure of the gas is $P. $ An identical vessel containing one mole of $He$ gas (molar mass $4)$ at temperature $2T$ has a pressure ofView Solution
- 5The specific heat of a gas at constant volume is $21.2\, J/mole/°C.$ If the temperature is increased by $1°C$ keeping the volume constant, the change in its internal energy will be ...... $J$View Solution
- 6The number of air molecules per $\mathrm{cm}^3$ increased from $3 \times 10^{19}$ to $12 \times 10^{19}$. The ratio of collision frequency of air molecules before and after the increase in number respectively is $.........$View Solution
- 7In the absence of intermolecular force of attraction, the observed pressure $P$ will beView Solution
- 8The relation between the gas pressure $P$ and average kinetic energy per unit volume $E$ isView Solution
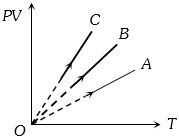
- 9$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
- 10The total kinetic energy of translatory motion of all the molecules of $5$ litres of nitrogen exerting a pressure $P$ is $3000 \,\,J$.View Solution
