Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]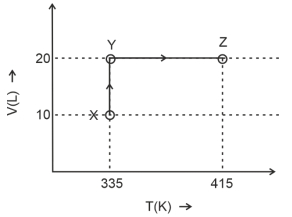
IIT 2024, Advanced
For ideal gas
$\Delta H=n C_P \Delta T$
$\because \quad C_P=C_V+R=12+8.3=20.3 J / K \text {-mole }$
$\therefore \Delta H=5 \times 20.3 \times(415-335)$
$\Delta H=8120 \text { Joule }$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 2View SolutionIn an isothermal change, an ideal gas obeys
- 3In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 4A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$View Solution
- 5$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 6View SolutionIn the following indicator diagram, the net amount of work done will be
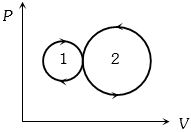
- 7Two samples $A$ and $B$ of a gas initially at the same pressure and temperature are compressed from volume $ V$ to $ V/2$ ($A$ isothermally and adiabatically). The final pressure of $ A$ isView Solution
- 8A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
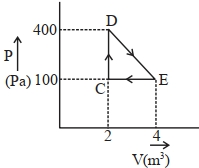
- 9An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
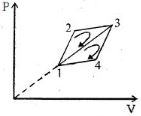
- 10The efficiency of a thermodynamic cycle $1-2-3- 1 ($see picture$)$ is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta ($ in $\%)$ of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution