A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$
JEE MAIN 2018, Medium
Given: Temperature of cold body, $T_2= 250\,K$ temperature of hot body; $T_1 = 300\, K$ Heat received, $Q_2= 500\, cal$ work done, $W =$ ?
Effociency$ = 1 - \frac{{{T_2}}}{{{T_1}}} = \frac{W}{{{Q_2} + W}}$
$ \Rightarrow 1 - \frac{{250}}{{300}} = \frac{W}{{{Q_2} + W}}$
$W = \frac{{{Q_2}}}{5} = \frac{{500 \times 4.2}}{5}\,\,J = 420\,J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
- 2Two moles of an ideal monoatomic gas occupies a volume $V$ at $27^o C$. The gas expands adiabatically to a volume $2\ V$. Calculate $(a)$ the final temperature of the gas and $(b)$ change in its internal energy.View Solution
- 3Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
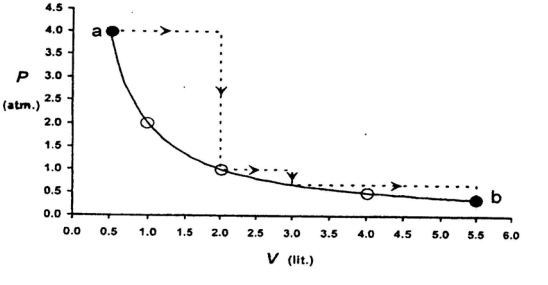
- 4One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ isView Solution
- 5A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
- 6View SolutionFor free expansion of the gas which of the following is true
- 7View SolutionFor adiabatic process, wrong statement is
- 8A mixture of gases at $STP$ for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{9}$ th of its original volume. The final temperature of mixture is .......... $^{\circ} C$View Solution
- 9A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 10Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
