During an adiabatic compression, $830\, J$ of work is done on $2\, moles$ of a diatomic ideal gas to reduce its volume by $50\%$. The change in its temperahture is nearly..... $K$ $(R\, = 8.3\, J\,K^{-1}\, mol^{-1} )$
JEE MAIN 2014, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
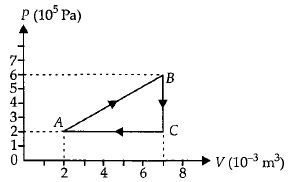
- 2A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 3For adiabatic processes $\left( {\gamma = \frac{{{C_p}}}{{{C_v}}}} \right)$View Solution
- 4One mole of an ideal gas at an initial temperature of $T\, K$ does $6\, R\, joules$ of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $\frac{5}{3}$ , the final temperature of gas will beView Solution
- 5Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$View Solution
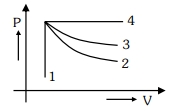
- 6An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among $1,2,3$ and $4$ isView Solution
- 7View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
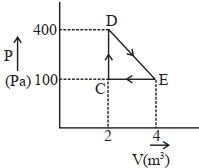
- 8A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
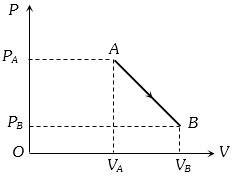
- 9An ideal gas is taken from point $A$ to the point $B,$ as shown in the $P-V$ diagram, keeping the temperature constant. The work done in the process isView Solution
- 10View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
