since gas undergoes isobaric process
$\Rightarrow \Delta \mathrm{Q}=\mathrm{n} \frac{7}{2} \mathrm{R\Delta\,T}=\frac{7}{2}({nR\Delta\,T})=35 \mathrm{J}$
Download our appand get started for free
Similar Questions
- 1An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
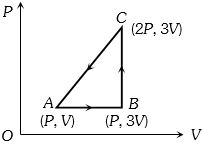
- 2An engine operating between the boiling and freezing points of water will haveView Solution
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
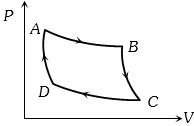
- 3Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
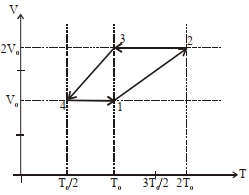
- 4One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$
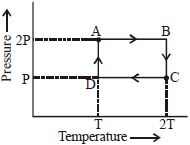
- 5An ideal monoatomic gas is taken through the thermodynamic states $A \to B \to C \to D$ via the paths shown in the figure. If $U_A, U_B, U_C$ and $U_D$ represent the internal energy of the gas in state $A, B\, C$ and $D$ respectively, then which of the following is not true?View Solution
- 6In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
- 7Starting at temperature $300\; \mathrm{K},$ one mole of an ideal diatomic gas $(\gamma=1.4)$ is first compressed adiabatically from volume $\mathrm{V}_{1}$ to $\mathrm{V}_{2}=\frac{\mathrm{V}_{1}}{16} .$ It is then allowed to expand isobarically to volume $2 \mathrm{V}_{2} \cdot$ If all the processes are the quasi-static then the final temperature of the gas (in $\left. \mathrm{K}\right)$ is (to the nearest integer)View Solution
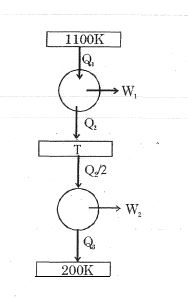
- 8Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 9View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
- 10Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
