During an experiment, an ideal gas is found to obey a condition $\frac{{{P^2}}}{\rho }$ = constant [$\rho =$ density of the gas]. The gas is initially at temperature $T,$ pressure $P$ and density $\rho$ . The gas expands such that density changes to $\rho/2$
Advanced
$\frac{P^{2}}{\rho}=$ constant
$P=p \frac{R T}{M}$ (Ideal gas equation)
$\Rightarrow \frac{p_{2}}{\rho}=\frac{P}{\rho}\left(\frac{\rho R T}{M}\right)=P T \frac{R}{M}=$ constant
$\therefore$ The graph of the above process on the $P-T$ diagram is hyperbola. For the above process
$\left(\frac{P_{2}}{\rho}\right)_{1}=\left(\frac{P^{2}}{\rho}\right)_{2} \Rightarrow \frac{P_{2}}{\rho}=\frac{P_{2}^{2}}{\rho / 2} \Rightarrow P_{2}=\frac{P}{\sqrt{2}} \ldots .(i)$
and
$P_{1} T_{1}=P_{2} T_{2} \Rightarrow P T=\frac{P}{\sqrt{2}} T_{2} \Rightarrow T_{2}=\sqrt{2} T \ldots .(ii)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
- 2Two moles of monoatomic gas is expanded from $(P_0, V_0)$ to $(P_0 , 2V_0)$ under isobaric condition. Let $\Delta Q_1$, be the heat given to the gas, $\Delta W_1$ the work done by the gas and $\Delta U_1$ the change in internal energy. Now the monoatomic gas is replaced by a diatomic gas. Other conditions remaining the same. The corresponding values in this case are $\Delta Q_2 , \Delta W_2 , \Delta U_2$ respectively, thenView Solution
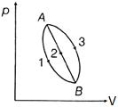
- 3An ideal gas of mass $m$ in a state $A$ goes to another state $B$ via three different processes as shown in figure. If $Q_{1}, Q_{2}$ and $Q_{3}$ denote the heat absorbed by the gas along the three paths, thenView Solution
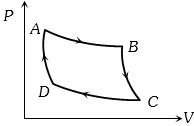
- 4Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
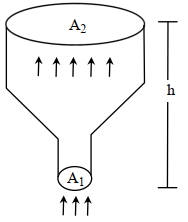
- 5An ideal gas of density $\rho=0.2 kg m ^{-3}$ enters a chimney of height $h$ at the rate of $\alpha=0.8 kg s ^{-1}$ from its lower end, and escapes through the upper end as shown in the figure. The cross-sectional area of the lower end is $A_1=0.1 m ^2$ and the upper end is $A_2=0.4 m ^2$. The pressure and the temperature of the gas at the lower end are $600 Pa$ and $300 K$, respectively, while its temperature at the upper end is $150 K$. The chimney is heat insulated so that the gas undergoes adiabatic expansion. Take $g=10 ms ^{-2}$ and the ratio of specific heats of the gas $\gamma=2$. Ignore atmospheric pressure.View Solution
Which of the following statement($s$) is(are) correct?
- 6The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
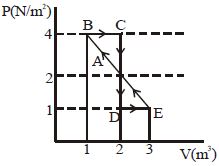
- 7One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
- 8Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
- 9A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process? $( J =4.18$ $Joules / cal )$ (in joule)View Solution
- 10View SolutionIf a system undergoes contraction of volume then the work done by the system will be
