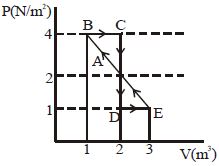
One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$

Medium
Work $=$ Area enclosed by curve
$\mathrm{W}=\mathrm{W}_{\mathrm{ABCD}}+\mathrm{W}_{\mathrm{DEAD}}$
$\mathrm{W}=1-\frac{1}{2}=\frac{1}{2} \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
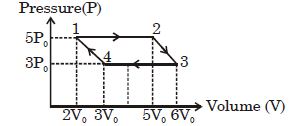
- 1An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
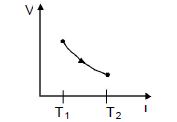
- 2The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution
- 3View SolutionIn the following indicator diagram, the net amount of work done will be
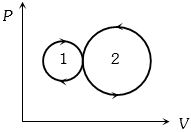
- 4Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
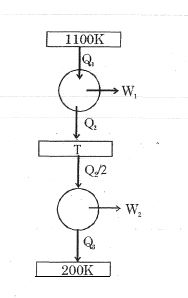
- 5If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
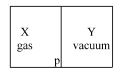
- 6A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?View Solution
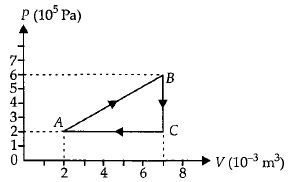
- 7A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 8$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
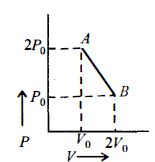
- 9Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
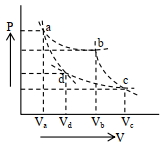
- 10Two different adiabatic paths for the same gas intersect two isothermal curves as shown in$P-V$ diagram. The relation between the ratio $\frac{V_a}{V_d}$ and the ratio $\frac{V_b}{V_c}$ is:View Solution