The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal to
AIIMS 2010, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas at a pressures of $1$ atmosphere and temperature of ${27^o}C$ is compressed adiabatically until its pressure becomes $8$ times the initial pressure, then the final temperature is ..... $^oC$ ($\gamma = 3/2$)View Solution
- 2One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 3A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$View Solution
- 4In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
- 5Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 6View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
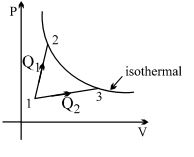
- 7A gas takes part in two processes in which it is heated from the same initial state $1$ to the same final temperature. The processes are shown on the $P-V$ diagram by the straight line $1-2$ and $1-3$. $2$ and $3$ are the points on the same isothermal curve. $Q_1$ and $Q_2$ are the heat transfer along the two processes. ThenView Solution
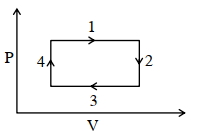
- 8An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
- 9View SolutionWhich of the following is a slow process
- 10A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
