A gas in container $A$ is in thermal equilibrium with another gas in container $B.$ both contain equal masses of the two gases in the respective containers. Which of the following can be true
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
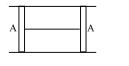
- 1A cylindrical tube of cross-sectional area $A$ has two air tight frictionless pistons at its two ends. The pistons are tied with a straight two ends. The pistons are tied with a straight piece of metallic wire. The tube contains a gas at atmospheric pressure $P_0$ and temperature $T_0$. If temperature of the gas is doubled then the tension in the wire isView Solution
- 2The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$View Solution
- 3One mole of an ideal monoatomic gas at temperature $T_0$ expands slowly according to the law $P/V$ = constant. If the final temperature is $2 \,\,T_0$, heat supplied to the gas is :View Solution
- 4The internal energy of $10\,g$ of nitrogen at $S.T.P$, is about ......... $J$View Solution
- 5Two vessels of the same volume contain the same gas at same temperature. If the pressure in the vessels be in the ratio of $1 : 2$, thenView Solution
- 6When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 7According to kinetic theory of gases,View Solution
$A$. The motion of the gas molecules freezes at $0^{\circ} C$
$B$. The mean free path of gas molecules decreases if the density of molecules is increased.
$C$. The mean free path of gas molecules increases if temperature is increased keeping pressure constant.
$D$. Average kinetic energy per molecule per degree of freedom is $\frac{3}{2} k_{B} T$ (for monoatomic gases)
Choose the most appropriate answer from the options given below
- 8An electron tube was sealed off during manufacture at a pressure of $1.2 \times {10^{ - 7}}$ $mm$ of mercury at $27°C.$ Its volume is $100\, cm^3$. The number of molecules that remain in the tube isView Solution
- 9$310\,J$ of heat is required to raise the temperature of $2\,moles$ of an ideal gas at constant pressure from $25\,^oC$ to $35\,^oC$ . The amount of heat required to raise the temperature of the gas through the same range at constant volume is .... $J$View Solution
- 10View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
