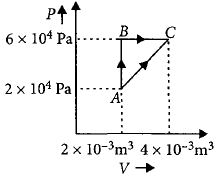
$\Delta {U_{ABC}} = \Delta {U_{AC}}$
$AB$ is isochoric process.
$\Delta {W_{AB}} = 0$
$\Delta {Q_{AB}} = \Delta {U_{AB}} = 400\,J$
$BC$ is isobaric process.
$\Delta {Q_{BC}} = \Delta {U_{BC}} + \Delta {W_{BC}}$
$100 = \Delta {U_{BC}} + 6 \times {10^4}\left( {4 \times {{10}^{ - 3}} - 2 \times {{10}^{ - 3}}} \right)$
$100 = \Delta {U_{BC}} + 12 \times 10$
$\Delta {U_{BC}} = 100 - 120 = - 20\,J$
$As,\,\Delta {U_{ABC}} = \Delta {U_{AC}}$
$\Delta {U_{AB}} + \Delta {U_{BC}} = \Delta {Q_{AC}} - \Delta {W_{AC}}$
$400 - 20 = \Delta {Q_{AC}} - (2 \times {10^4} \times 2 \times {10^{ - 3}} + \frac{1}{2} \times $
$2 \times {10^{ - 3}} \times 4 \times {10^4})$
$\Delta {Q_{AC}} = 460J$
Download our appand get started for free
Similar Questions
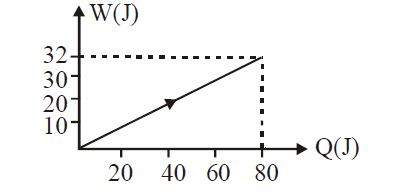
- 1View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
- 2A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 3Two moles of an ideal monoatomic gas at ${27^o}C$ occupies a volume of $V.$ If the gas is expanded adiabatically to the volume $2V,$ then the work done by the gas will be ....... $J$ $[\gamma = 5/3,\,R = 8.31J/mol\,K]$View Solution
- 4A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 5If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
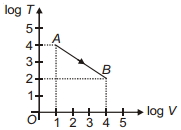
- 6Figure shows, the adiabatic curve on a $\log T$ and log $V$ scale performed on ideal gas. The gas is ............View Solution
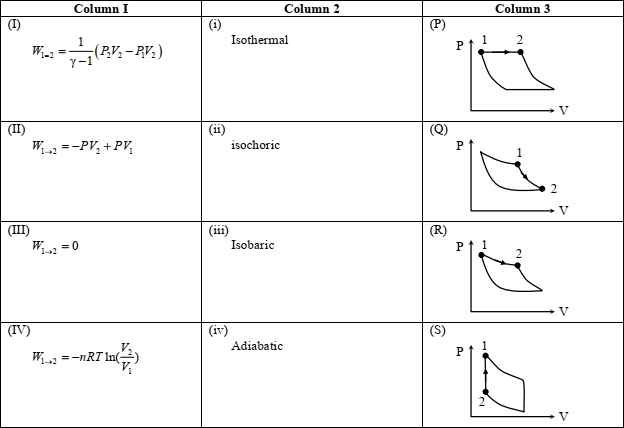
- 7An ideal gas is undergoing a cyclic thermodynamic process in different ways as shown in the corresponding $P$ $V$ diagrams in column $3$ of the table. Consider only the path from state $1$ to $2 . W$ denotes the corresponding work done on the system. The equations and plots in the table have standard notations as used in thermodynamic processes. Here $\gamma$ is the ratio of heat capacities at constant pressure and constant volume. The number of moles in the gas is $n$.View Solution
(image)
($1$) Which of the following options is the only correct representation of a process in which $\Delta U=\Delta Q-P \Delta V$ ?
$[A] (II) (iv) (R)$ $[B] (II) (iii) (P)$ $[C] (II) (iii) (S)$ $[D] (III) (iii) (P)$
($2$) Which one of the following options is the correct combination?
$[A] (III) (ii) (S)$ $[B] (II) (iv) (R)$ $[C] (II) (iv) (P)$ $[D] (IV) (ii) (S)$
($3$) Which one of the following options correctly represents a thermodynamic process that is used as a correction in the determination of the speed of sound in an ideal gas?
$[A] (III) (iv) (R)$ $[B] (I) (ii)$ $(\mathrm{Q})$ $[C] (IV) (ii) (R)$ $[D] (I) (iv) (Q)$
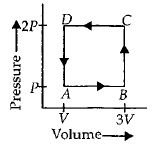
- 8A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
- 9The amount of heat needed to raise the temperature of $4\, moles$ of a rigid diatomic gas from $0^{\circ} {C}$ to $50^{\circ} {C}$ when no work is done is ......${R}$ ($R$ is the universal gas constant)View Solution
- 10View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
