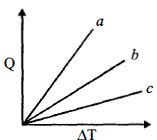
Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond to

JEE MAIN 2013, Medium
On giving same amount of heat at constant pressure, there is no change in temperature for mono, dia and polyatomic.
$(\Delta \mathrm{Q})_{\mathrm{P}}=\mu \mathrm{C}_{\mathrm{p}} \Delta \mathrm{T}\left(\mu=\begin{array}{l}{\text { No. of molecules }} \\ {\text { Avogedro's na }}\end{array}\right)$
or $\Delta \mathrm{T} \propto \frac{1}{\text { no of molecules }}$
$(\Delta \mathrm{Q})_{\mathrm{P}}=\mu \mathrm{C}_{\mathrm{p}} \Delta \mathrm{T}\left(\mu=\begin{array}{l}{\text { No. of molecules }} \\ {\text { Avogedro's na }}\end{array}\right)$
or $\Delta \mathrm{T} \propto \frac{1}{\text { no of molecules }}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionFour mole of hydrogen, two mole of helium and one mole of water vapour form an ideal gas mixture. What is the molar specific heat at constant pressure of mixture?
- 2A gas has $n$ degrees of freedom. The ratio of specific heat of gas at constant volume to the specific heat of gas at constant pressure will be.View Solution
- 3$P-V$ diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will beView Solution
- 4A cylinder contains $10\, kg$ of gas at pressure of ${10^7}\,\,N/{m^2}.$ The quantity of gas taken out of the cylinder, if final pressure is $2.5 \times {10^6}\,N/{m^2},$ will be ..... $kg$ (Temperature of gas is constant)View Solution
- 5$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?View Solution
- 6A cylinder of fixed capacity $44.8$ litre. contains a monatomic gas at standard temperature and pressure. The amount of heat required to cylinder by $10^o C$ will be. ($R=$ universal gas constant)View Solution
- 7In the two vessels of same volume, atomic hydrogen and helium at pressure $1\, atm$ and $2\, atm$ are filled. If temperature of both the samples is same, then average speed of hydrogen atoms $ < {C_H} > $ will be related to that of helium $ < {C_{He}} > $ asView Solution
- 8Statement$-1 :$ Internal energy of gas $U = nC_VT$ is due to random motion of gas molecules.View Solution
Statement$-2 :$ A container is moving with speed $v$. It is suddenly stopped by a force, temperature of gas increases. - 9View SolutionAt absolute zero temperature, pressure of a gas will be
- 10The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
