$P-V$ diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
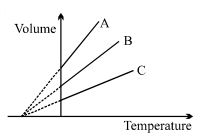
- 1The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2 \,\,m$ at a pressure $2P$ is given by the straight lineView Solution
- 2If pressure of a gas contained in a closed vessel is increased by $0.4\%$ when heated by $1°C,$ the initial temperature must beView Solution
- 3For a gas, the $r.m.s.$ speed at $800\, K$ isView Solution
- 4Consider a $1\, c.c.$ sample of air at absolute temperature ${T_0}$ at sea level and another $1 cc$ sample of air at a height where the pressure is one-third atmosphere. The absolute temperature $T$ of the sample at that height isView Solution
- 5View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
- 6If the r.m.s. speed of chlorine molecule is $490\,m / s$ at $27^{\circ}\,C$, the r.m.s. speed of argon molecules at the same temperature will be $......\,m/s$ (Atomic mass of argon $=39.9\,u$, molecular mass of chlorine $=70.9\,u )$View Solution
- 7The temperature of argon, kept in a vessel, is raised by $1^\circ C$ at a constant volume. The total heat supplied to the gas is a combination of translational and rotational energies. Their respective shares areView Solution
- 8The Mean Free Path $\ell$ for a gas molecule depends upon diameter, $d$ of the molecule asView Solution
- 9An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):View Solution
- 10A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:View Solution
