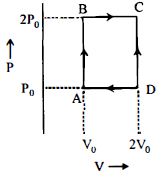
First law of thermodynamics is a special case of
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn adiabatic expansion of a gas
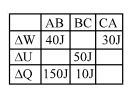
- 2A student records $\Delta Q, \Delta U \& \Delta W$ for a thermodynamic cycle $A$ $\rightarrow$ $B \rightarrow C \rightarrow A$. Certain entries are missing. Find correct entry in following options.View Solution
- 3When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
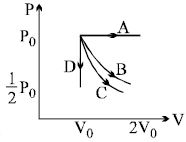
- 4The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process(es) does the temperature of the gas decrease ?View Solution
- 5A gas is compressed at a constant pressure of $50N/{m^2}$ from a volume of $10{m^3}$ to a volume of $4{m^3}$. Energy of $100 J$ then added to the gas by heating. Its internal energy isView Solution
- 6One mole of an ideal gas at $300 \mathrm{~K}$ in thermal contact with surroundings expands isothermally from $1.0 \mathrm{~L}$ to $2.0 \mathrm{~L}$ against a constant pressure of $3.0 \mathrm{~atm}$. In this process, the change in entropy of surroundings $\left(\Delta S_{\text {surr }}\right)$ in $\mathrm{J} \mathrm{K}^{-1}$ is $(1 \mathrm{~L} \mathrm{~atm}=101.3 \mathrm{~J})$View Solution
- 7A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
- 8View SolutionFor free expansion of the gas which of the following is true
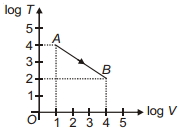
- 9Figure shows, the adiabatic curve on a $\log T$ and log $V$ scale performed on ideal gas. The gas is ............View Solution
- 10An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)View Solution