Following statements are given :
$(1)$ The average kinetic energy of a gas molecule decreases when the temperature is reduced.
$(2)$ The average kinetic energy of a gas molecule increases with increase in pressure at constant temperature.
$(3)$ The average kinetic energy of a gas molecule decreases with increases in volume.
$(4)$ Pressure of a gas increases with increase in temperature at constant pressure.
$(5)$ The volume of gas decreases with increase in temperature.
Choose the correct answer from the options given below:
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A perfect gas at $27\,^oC$ is heated at constant pressure so as to double its volume. The final temperature of the gas will be, close to ...... $^oC$View Solution
- 2A thermally insulated rigid container of $1 \,L$ volume contains a diatomic ideal gas at room temperature. A small paddle installed inside the container is rotated from the outside, such that the pressure rises by $10^{5} \,Pa$. The change in internal energy is close to ............... $J$View Solution
- 3View SolutionThe degrees of freedom of a triatomic gas is
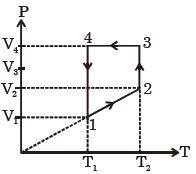
- 4A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 5At $NTP$ the mass of one litre of air is $1.293\, gm.$ The value of specific gas constant will be ...... $J/K-gm$View Solution
- 6View SolutionA cylinder rolls without slipping down an inclined plane, the number of degrees of freedom it has, is
- 7Energy of all molecules of a monoatomic gas having a volume $V$ and pressure $P$ is $\frac{3}{2}PV$. The total translational kinetic energy of all molecules of a diatomic gas as the same volume and pressure isView Solution
- 8The mean free path of gas molecules depends on $(d =$ molecular diameter$)$View Solution
- 9At temperature $T,$ the $r.m.s.$ speed of helium molecules is the same as $r.m.s.$ speed of hydrogen molecules at normal temperature and pressure. The value of $T$ is ....... $^oC$View Solution
- 10If an ideal gas has volume $V$ at $27°C$ and it is heated at a constant pressure so that its volume becomes $1.5V.$ Then the value of final temperature will be ....... $^oC$View Solution
