The degrees of freedom of a triatomic gas is
AIPMT 1999, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
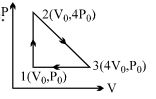
- 2One mole of an ideal diatomic gas is taken through the cycle as shown in the figure.View Solution
$1 \rightarrow 2 :$ isochoric process
$2 \rightarrow 3 :$ straight line on $P-V$ diagram
$3 \rightarrow 1 :$ isobaric process
The average molecular speed of the gas in the states $1, 2$ and $3$ are in the ratio
- 3A gas at the temperature $250\, K$ is contained in a closed vessel. If the gas is heated through $1\,K,$ then the percentage increase in its pressure will be .... $\%$View Solution
- 4Two vessels $A$ and $B$ are of the same size and are at same temperature. A contains $1 \mathrm{~g}$ of hydrogen and $B$ contains $1 \mathrm{~g}$ of oxygen. $\mathrm{P}_{\mathrm{A}}$ and $\mathrm{P}_{\mathrm{B}}$ are the pressures of the gases in $A$ and $\mathrm{B}$ respectively, then $\frac{\mathrm{P}_{\mathrm{A}}}{\mathrm{P}_{\mathrm{B}}}$ is:View Solution
- 5An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true$?$View Solution
$(A)$ the mean free path of the molecules decreases.
$(B)$ the mean collision time between the molecules decreases.
$(C)$ the mean free path remains unchanged.
$(D)$ the mean collision time remains unchanged.
- 6A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 7The root mean square velocity of the molecules in a sample of helium is $(5/7)^{th}$ that of the molecules in a sample of hydrogen. If the temperature of hydrogen sample is $0°C,$ then the temperature of the helium sample is aboutView Solution
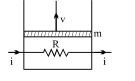
- 8A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
- 9If the root mean square velocity of the molecules of hydrogen at $NTP$ is $1.84\, km/s$. Calculate the root mean square velocity of oxygen molecule at $NTP$, molecular weight of hydrogen and oxygen are $2$ and $32$ respectively ....... $km/sec$View Solution
- 10View SolutionA closed compartment containing gas is moving with some acceleration in horizontal direction. Neglect effect of gravity. Then the pressure in the compartment is
