Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$View Solution
- 2View SolutionA container that suits the occurrence of an isothermal process should be made of
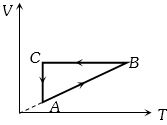
- 3A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
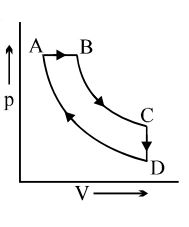
- 4A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 5During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
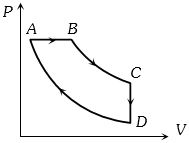
- 6A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 7$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
- 8A diatomic gas undergoes a process represented by $PV ^{1.3}=$ constant. Choose the incorrect statementView Solution
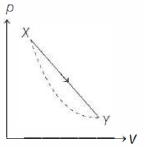
- 9In the $p-V$ diagram below, the dashed curved line is an adiabat.For a process that is described by a straight line joining two points $X$ and $Y$ on the adiabat (solid line in the diagram) heat is (Hint consider the variation in temperature from $X$ to $Y$ along the straight line)View Solution
- 10In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will beView Solution
