In the $p-V$ diagram below, the dashed curved line is an adiabat.For a process that is described by a straight line joining two points $X$ and $Y$ on the adiabat (solid line in the diagram) heat is (Hint consider the variation in temperature from $X$ to $Y$ along the straight line)
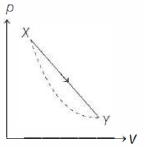

KVPY 2013, Advanced
(c)
As we move along line $X Y$, it cuts a isotherm at higher temperature $\left(T_2 > T_3\right)$
$\left(T_2 > T_3\right)$ but then from $Z$ to $Y$ line moves to a low temperature isotherm. So, heat is absorbed upto $Z$ and then it is released from $Z$ to $Y$.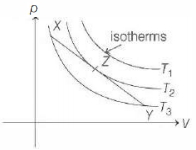
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
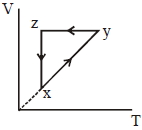
- 1A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 2$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 3View SolutionIn which thermodynamic process, volume remains same
- 4A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
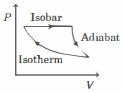
- 5The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
- 6A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
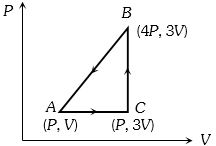
- 7A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
- 8$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
- 9In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 10A closed container contains a homogeneous mixture of two moles of an ideal monatomic gas $(\gamma=5 / 3)$ and one mole of an ideal diatomic gas $(\gamma=7 / 5)$. Here, $\gamma$ is the ratio of the specific heats at constant pressure and constant volume of an ideal gas. The gas mixture does a work of $66$ Joule when heated at constant pressure. The change in its internal energy is. . . . . . .Joule.View Solution
