A diatomic gas undergoes a process represented by $PV ^{1.3}=$ constant. Choose the incorrect statement
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 2Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
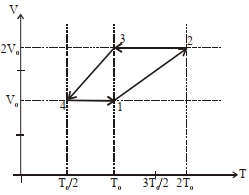
- 3One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$
- 4The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
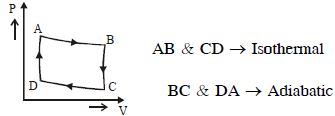
- 5View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 6An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
- 7Two moles of monoatomic gas is expanded from $(P_0, V_0)$ to $(P_0 , 2V_0)$ under isobaric condition. Let $\Delta Q_1$, be the heat given to the gas, $\Delta W_1$ the work done by the gas and $\Delta U_1$ the change in internal energy. Now the monoatomic gas is replaced by a diatomic gas. Other conditions remaining the same. The corresponding values in this case are $\Delta Q_2 , \Delta W_2 , \Delta U_2$ respectively, thenView Solution
- 8During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
- 9During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?View Solution
- 10One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
