For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$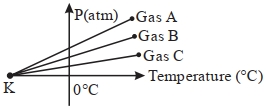
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In Vander Waal’s equation $a$ and $b$ represent $\left( {P + \frac{a}{{{V^2}}}} \right)\,(V - b) = RT$View Solution
- 2The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
- 3Statement$-1$ : Real gas approaches ideal gas behaviour for low pressures and high temperatures.View Solution
statement$-2 $: At low pressure, density of gas is very low. - 4View SolutionMolecular motion shows itself as
- 5View SolutionThe absolute zero is the temperature at which
- 6By what factor the $r.m.s.$ velocity will change, if the temperature is raised from $27^\circ C$ to $327^\circ C$View Solution
- 7A closed cylindrical vessel contains $N$ moles of an ideal diatomic gas at a temperature $T$. On supplying heat, temperature remains same, but $n$ moles get dissociated into atoms. The heat supplied is .........View Solution
- 8For a gas, the $r.m.s.$ speed at $800\, K$ isView Solution
- 9Find the ratio of specific heat at constant pressure to the specific heat constant volume for $N{H_3}$View Solution
- 10The average translational energy and the $r.m.s.$ speed of molecules in a sample of oxygen gas at $300 K$ are $6.21 \times {10^{ - 21}}\,J$ and $484\, m/s$ respectively. The corresponding values at $600 K$ are nearly (assuming ideal gas behaviour)View Solution
