In Vander Waal’s equation $a$ and $b$ represent $\left( {P + \frac{a}{{{V^2}}}} \right)\,(V - b) = RT$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas at temperature $T_0$ expands slowly according to the law $P/V$ = constant. If the final temperature is $2 \,\,T_0$, heat supplied to the gas is :View Solution
- 2For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
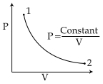
- 3View SolutionMoon has no atmosphere because
- 4$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
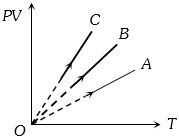
- 5View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
- 6If one mole of the polyatomic gas is having two vibrational modes and $\beta$ is the ratio of molar specific heats for polyatomic gas $\left(\beta=\frac{ C _{ P }}{ C _{ v }}\right)$ then the value of $\beta$ is:View Solution
- 7A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:View Solution
- 8The molar heat capacities of a mixture of two gases at constant volume is $13 R / 6$. The ratio of number of moles of the first gas to the second is $1: 2$. The respective gases may beView Solution
- 9View SolutionWhich one the following graphs represents the behaviour of an ideal gas
- 10View SolutionSaturated vapour is compressed to half its volume without any change in temperature, then the pressure will be
