From the following statements, concerning ideal gas at any given temperature $T,$ select the correct one(s)
IIT 1995, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T,$ the mass of each molecule is $m.$ The expression for the density of gas is ($k =$ Boltzmann’s constant)View Solution
- 2At $0°C $ the density of a fixed mass of a gas divided by pressure is $x.$ At $100°C,$ the ratio will beView Solution
- 3Volume-temperature graph at atmospheric pressure for a monoatomic gas $(V\,{\rm{in }}{m^3},\,\,T\,{\rm{in}}{{\rm{ }}^{\rm{o}}}C)$ isView Solution
- 4At $27°C$ temperature, the kinetic energy of an ideal gas is ${E_1}$. If the temperature is increased to $327°C,$ then kinetic energy would beView Solution
- 5$Assertion :$ One mole of any substance at any temperature or volume always contains $6.02\times10^{23}$ molecules.View Solution
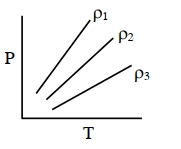
$Reason :$ One mole of a substance always refers to $S.T.P.$ conditions. - 6$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
- 7If a mixture of $28\,\, g$ of Nitrogen, $4 \,\,g$ of Hydrogen and $8 \,\,gm$ of Helium is contained in a vessel at temperature $400 \,\,K$ and pressure $8.3 \times 10^5 \,\,Pa$, the density of the mixture will be :View Solution
- 8View SolutionFor ideal gas, which statement is not true
- 9View SolutionSelect the appropriate property of an ideal gas
- 10An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true$?$View Solution
$(A)$ the mean free path of the molecules decreases.
$(B)$ the mean collision time between the molecules decreases.
$(C)$ the mean free path remains unchanged.
$(D)$ the mean collision time remains unchanged.
