A sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T,$ the mass of each molecule is $m.$ The expression for the density of gas is ($k =$ Boltzmann’s constant)
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $7\, gm N _{2}$ is mixed with $20\, gm$ $Ar$, there $C _{ p } / C _{ v }$ of mixture will beView Solution
- 2Let $\bar v,\;{v_{rms}}$ and ${v_p}$ respectively denote the mean speed, root mean square speed and most probable speed of the molecules in an ideal monoatomic gas at absolute temperature $T.$ The mass of a molecule is $m.$ ThenView Solution
- 3Two moles of an ideal gas with $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}=\frac{5}{3}$ are mixed with $3$ moles of another ideal gas with $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}=\frac{4}{3} .$ The value of $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}$ for the mixture isView Solution
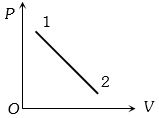
- 4A volume $V$ and pressure $P$ diagram was obtained from state $1$ to state $2$ when a given mass of a gas is subjected to temperature changes. During this process the gas isView Solution
- 5Let $A$ and $B$ the two gases and given : $\frac{{{T_A}}}{{{M_A}}} = 4.\frac{{{T_B}}}{{{M_B}}};$ where $T$ is the temperature and M is molecular mass. If ${C_A}$ and ${C_B}$ are the $r.m.s. $ speed, then the ratio $\frac{{{C_A}}}{{{C_B}}}$ will be equal toView Solution
- 6View SolutionThe degrees of freedom of a triatomic gas is
- 7For a gas, the $r.m.s.$ speed at $800\, K$ isView Solution
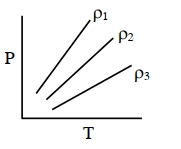
- 8$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
- 9A balloon carries a total load of $185\; {kg}$ at normal pressure and temperature of $27^{\circ} {C}$. What load will the balloon carry on rising to a height at which the barometric pressure is $45\; {cm}$ of ${Hg}$ and the temperature is $-7^{\circ} {C}$. Assuming the volume constant? (in ${kg}$)View Solution
- 10View SolutionAt absolute zero temperature, pressure of a gas will be
