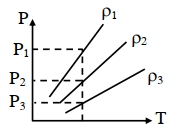
$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :

JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ The root mean square and most probable speeds of the molecules in a gas are the same.View Solution
$Reason :$ The Maxwell distribution for the speed of molecules in a gas in symmetrical. - 2A perfect gas at $27\,^oC$ is heated at constant pressure so as to double its volume. The final temperature of the gas will be, close to ...... $^oC$View Solution
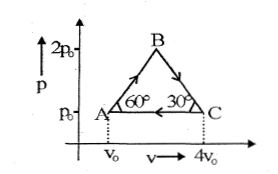
- 3Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
- 4$Assertion :$ For a gas atom the number of degrees of freedom is $3$.View Solution
$Reason :$ $\frac{{{C_P}}}{{{C_V}}} = \gamma $ - 5By what factor the $r.m.s.$ velocity will change, if the temperature is raised from $27^\circ C$ to $327^\circ C$View Solution
- 6$125\, ml$ of gas $A$ at $0.60$ atmosphere and $150\, ml$ of gas $B$ at $0.80$ atmosphere pressure at same temperature is filled in a vessel of $1$ litre volume. What will be the total pressure of mixture at the same temperature ............... $\mathrm{atmosphere}$View Solution
- 7A partition divides a container having insulated walls into two compartments $I$ and $II$. the same gas fills the two compartments. The ratio of the number of molecules in compartments $I$ and $II$ isView Solution

- 8If the volume of the gas containing $n$ number of molecules is $V,$ then the pressure will decrease due to force of intermolecular attraction in the proportionView Solution
- 9A sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T,$ the mass of each molecule is $m.$ The expression for the density of gas is ($k =$ Boltzmann’s constant)View Solution
- 10The equation of state of a gas is given by $\left( {P + \frac{{a{T^2}}}{V}} \right)\,{V^c} = (RT + b)$, where $a, b, c$ and $R$ are constants. The isotherms can be represented by $P = A{V^m} - B{V^n}$, where $A$ and $B$ depend only on temperature thenView Solution