Given below are two statements :
Statement $(I)$ : The mean free path of gas molecules is inversely proportional to square of molecular diameter.
Statement $(II)$ : Average kinetic energy of gas molecules is directly proportional to absolute temperature of gas.
In the light of the above statements, choose the correct answer from the option given below:
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $\alpha$ moles of a monoatomic gas are mixed with $\beta$ moles of a polyatomic gas and mixture behaves like diatomic gas, then [neglect the vibrational mode of freedom]View Solution
- 2A flask contains argon and oxygen in the ratio of $3: 2$ in mass and the mixture is kept at $27^{\circ} C$. The ratio of their average kinetic energy per molecule respectively will be ...........View Solution
- 3A mixture of hydrogen and oxygen has volume $2000 \; cm ^{3}$, temperature $300 \; K$, pressure $100 \; kPa$ and mass $0.76 \; g$ The ratio of number of moles of hydrogen to number of moles of oxygen in the mixture will beView Solution
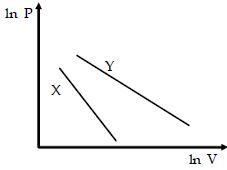
- 4For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution
- 5The relation between the gas pressure $P$ and average kinetic energy per unit volume $E$ isView Solution
- 6A very tall vertical cylinder is filled with a gas of molar mass $M$ under isothermal conditions at temperature $T.$ The density and pressure of the gas at the base of the container is $\rho_0$ and $p_0$, respectively Choose the correct statement(s)View Solution
- 7Molar specific heat at constant volume is ${C_v}$ for a monoatomic gas isView Solution
- 8The volume of a gas will be double of what it is at $0°C$ (pressure remaining constant) atView Solution
- 9View SolutionA container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is not ideal, then
- 10A volume $V$ and pressure $P$ diagram was obtained from state $1$ to state $2$ when a given mass of a gas is subjected to temperature changes. During this process the gas isView Solution