The volume of a gas will be double of what it is at $0°C$ (pressure remaining constant) at
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
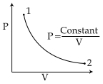
- 1For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
- 2View SolutionFor matter to exist simultaneously in gas and liquid phases
- 3View SolutionIn which of the following gas the root mean square velocity will be minimum (at const. temperature)
- 4A hot air balloon with a payload rises in the air. Assume that the balloon is spherical in shape with diameter of $11.7 \,m$ and the mass of the balloon and the payload (without the hot air inside) is $210 \,kg$. Temperature and pressure of outside air are $27^{\circ} C$ and $1 atm =10^5 \,N / m ^2$, respectively. Molar mass of dry air is $30 \,g$. The temperature of the hot air inside is close to .......... $^{\circ} C$ [The gas constant, $R=8.31 \,JK ^{-1} mol ^{-1}$ ]View Solution
- 5$0.056 \,kg$ of Nitrogen is enclosed in a vessel at a temperature of $127\,^{\circ} C$. The amount of heat required to double the speed of its molecules is k cal. (Take $R =2$ $cal \,mole$ $^{-1} K ^{-1}$ )View Solution
- 6$14 \,g$ of $CO$ at $27^{\circ} C$ is mixed with $16 g$ of $O _2$ at $47^{\circ} C$. The temperature of mixture is .......... $^{\circ} C$ (vibration mode neglected)View Solution
- 7The equation of state corresponding to $8 \,g$ of ${O_2}$ isView Solution
- 8View SolutionThe specific heat of a gas in a polytropic process is given by
- 9To what temperature should the hydrogen at room temperature $(27°C)$ be heated at constant pressure so that the $R.M.S.$ velocity of its molecules becomes double of its previous value ....... $^oC$View Solution
- 10At what temperature the molecules of nitrogen will have the same $r.m.s.$ velocity as the molecules of oxygen at $127°C$ ....... $^oC$View Solution
