How will you separate a mixture of kerosene oil and water? Explain with the help of a labeled diagram
The mixture of water and kerosene is put in separating funnel and allowed to stand for some time. The mixture separates into two layers according to the densities of water and kerosene. Water is heavier than kerosene. So, water forms lower layer while kerosene forms upper layer. On opening the stop clock of separating funnel, the lower layer of water comes out first and collected in beaker leaving behind kerosene. 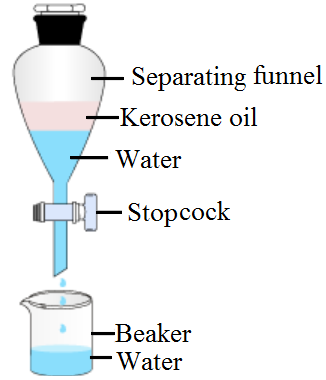 Separation of kerosene oil and water mixture by separating funnel.
Separation of kerosene oil and water mixture by separating funnel.
 Separation of kerosene oil and water mixture by separating funnel.
Separation of kerosene oil and water mixture by separating funnel.Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionList five characteristics by which compounds can be distinguished from mixtures.
- 2View SolutionA mixture contains water, kerosene and sand. How will you separate this mixture?
- 3How would you prepare a saturated solution of sodium chloride in water at $25^\circ C?$ What will happen if this solution is cooled to $10^\circ C?$View Solution
- 4View SolutionDiscuss the method of separating a mixture containing chalk powder, iron fillings and naphthalene.
- 5View SolutionWhat is fractional distillation? What is the use of fractionating column in fractional distillation?
- 6How can you obtain pure water from a salt-water mixture $($or salt-solution$)?$ Draw a meat and labelled diagram of the apparatus you would use to obtain pure water from a salt-water mixture $($or salt-solution$).$View Solution
- 7Iron powder and sulphur powder were mixed together and divided into two parts $A$ and $B.$ When part $A$ was heated strongly over a burner, then a substance $C$ was formed. The part $B$ was, however, not heated at all. When dilute hydrochloric acid was added to substance $C,$ then gas $D$ was evolved and when dilute hydrochloric acid was added to part $B$ then gas $E$ was evolved.View Solution
- What type of substance is $B?$
- What type of substance is $C?$
- Name the gas $(i)\ D,$ and $(ii)\ E?$
- Write one characteristic property of gas $D.$
- 8$A , B$ and $C$ are all liquids. Liquid $A$ has a comparatively low boiling point. On heating, liquid a vaporizes completely without leaving behind any residue. Liquid $A$ is being used increasingly as a fuel in motor vehicles either alone or by mixing with petrol. Liquid $B$ has a very high boiling point. It also vaporizes completely on heating, without leaving any residue. Liquid $B$ is a conductor of electricity and used in making thermometers. Liquid $c$ has a moderate boiling point. On heating, liquid $C$ vaporises leaving behind a white solid $D$ which is used in cooking vegetables. The condensation of vapours from $C$ give a liquid $E$ which turns anhydrous $CuSO _4$ to blue.View Solution
a. Which liquid could be an element$?$ Name this element.
b. Which liquid could be a mixture$?$ Name this mixture.
c. Which liquid could be a compound$?$ Name this compound.
d. What could the solid $D$ be$?$
e. What do you think is liquid $E$ ? - 9View SolutionHow is water purified on a large scale at water works? Explain with the help of a labelled diagram. Name the substance which is added to kill germs in the drinking water supply?
- 10View SolutionDifferentiate between a saturated and an unsaturated solution. How will you test whether a given solution is saturated or not?
