If a gas is heated at constant pressure, its isothermal compressibility
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $\gamma=3 / 2$. The work done by the gas in the process is : $(\mu=1 \mathrm{~mole})$View Solution
- 2A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
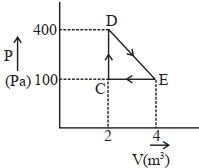
- 3A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
- 4A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 5View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 6View SolutionThe internal energy of an ideal gas depends upon
- 7A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$View Solution
- 8An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$View Solution
- 9Two cylinders $A$ and $B$ of equal capacity are connected to each other via a stop cock. A contains an Ideal gas at standard temperature and pressure. $B$ is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is :View Solution
- 10$Assertion :$ When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.View Solution
$Reason :$ Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours.
