A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$
JEE MAIN 2021, Medium
$\eta=\frac{ T _{2}}{ T _{1}}=\frac{ Q _{2}}{ Q _{1}}=\frac{ Q _{1}- W }{ Q _{1}} \quad\left(\because W = Q _{1}- Q _{2}\right)$
$\frac{400}{800}=1-\frac{ W }{ Q _{1}}$
$\frac{ W }{ Q _{1}}=1-\frac{1}{2}=\frac{1}{2}$
$Q _{1}=2 W =2400 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $\gamma = 2.5$ and volume is equal to $\frac{1}{8}$ times to the initial volume then pressure $P' $ is equal to (Initial pressure $= P$)View Solution
- 2A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
- 3In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20\,J$ of heat and $8\,J$ of work is done on the gas. If the initial internal energy of the gas was $30\,J$. The final internal energy will be...... $J$View Solution
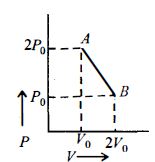
- 4$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
- 5For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 6Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
- 7Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
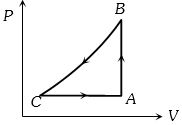
- 8A sample of an ideal gas is taken through a cycle a shown in figure. It absorbs $50J$ of energy during the process $AB$, no heat during $BC$, rejects $70J$ during $CA.$ $40J$ of work is done on the gas during $BC$. Internal energy of gas at $A$ is $1500J$, the internal energy at $C$ would be ........ $J$View Solution
- 9Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 10If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
