If number of molecules of ${H_2}$ are double than that of ${O_2}$, then ratio of kinetic energy of hydrogen and that of oxygen at $300 \,K$ is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The number of gas molecules striking per second per square metre of the top surface of a table placed in a room at $20^{\circ} C$ and 1 atmospheric pressure is of the order of $\left(k_{B}=1.4 \times 10^{-23} \,JK ^{-1}\right.$ and the average mass of an air molecule is $5 \times 10^{-27} \,kg$ )View Solution
- 2The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 3The $r.m.s.$ speed of the molecules of a gas in a vessel is $400$ $m{s^{ - 1}}$. If half of the gas leaks out, at constant temperature, the $r.m.s.$ speed of the remaining molecules will be ..... $ms^{-1}$View Solution
- 4The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 5View SolutionWhich law states that effect of pressure is same for all portion
- 6Two moles of a monoatomic ideal gas is confined in a container and is heated such that its temperature increases by $10\,^oC$. The approximate change in its internal energy is ..... $J$. $(R = 8.31\, J/mole-K)$View Solution
- 7A thermally insulated rigid container of $1 \,L$ volume contains a diatomic ideal gas at room temperature. A small paddle installed inside the container is rotated from the outside, such that the pressure rises by $10^{5} \,Pa$. The change in internal energy is close to ............... $J$View Solution
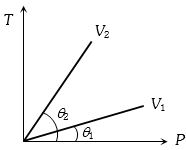
- 8From the following $P-T$ graph what interference can be drawnView Solution
- 9A vertical closed cylinder is separated into two parts by a frictionless piston of mass $m$ and of negligible thickness. The piston is free to move along the length of the cylinder .The length of the cylinder above the piston is $l_1,$ and that below the piston is $l_2,$ such that $l_1 > l_2.$ Each part of the cylinder contains $n$ moles of an ideal gas at equal temperature $T.$ If the piston is stationary, its mass, $m,$ will be given by: ( $R$ is universal gas constant and $g$ is the acceleration due to gravity)View Solution
- 10A cylinder with fixed capacity of $67.2\, lit$ contains helium gas at $STP$. The amount of heat needed to raise the temperature of the gas by $20\,^oC$ is ..... $J$ [Given that $R = 8.31\, J\, mol^{-1}\, K^{-1}$]View Solution
