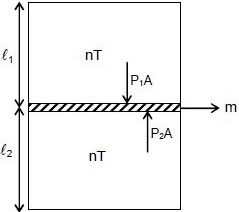
A vertical closed cylinder is separated into two parts by a frictionless piston of mass $m$ and of negligible thickness. The piston is free to move along the length of the cylinder .The length of the cylinder above the piston is $l_1,$ and that below the piston is $l_2,$ such that $l_1 > l_2.$ Each part of the cylinder contains $n$ moles of an ideal gas at equal temperature $T.$ If the piston is stationary, its mass, $m,$ will be given by: ( $R$ is universal gas constant and $g$ is the acceleration due to gravity)
JEE MAIN 2019, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two bulbs of identical volumes connected by a small capillary are initially filled with an ideal gas at temperature $T$. Bulb $2$ is heated to maintain a temperature $2 T$, while bulb $1$ remains at temperature $T$. Assume throughout that the heat conduction by the capillary is negligible. Then, the ratio of final mass of the gas in bulb $2$ to the initial mass of the gas in the same bulb is close toView Solution
- 2View SolutionOn absolute temperature, the kinetic energy of the molecules
- 3The gas having average speed four times as that of $S{O_2}$ (molecular mass $64$) isView Solution
- 4The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
- 5Air is filled in a bottle at atmospheric pressure and it is corked at $35°C.$ If the cork can come out at $3$ atmospheric pressure than upto what temperature should the bottle be heated in order to remove the cork ...... $^oC$View Solution
- 6An ideal gas occupies a volume of $2\, m^3$ at a pressure of $3\times10^6\, Pa$. The energy of the gas isView Solution
- 7At which temperature the velocity of $O_2$ molecules will be equal to the velocity of $N_2$ molecules at $0\,^oC$ ....... $^oC$View Solution
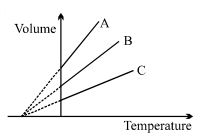
- 8The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2 \,\,m$ at a pressure $2P$ is given by the straight lineView Solution
- 9Equation of gas in terms of pressure $(P),$ absolute temperature $(T)$ and density $(d)$ isView Solution
- 10Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2 : 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4 : 3$. The ratio of their densities is :-View Solution