The internal energy of an ideal gas increases during an isothermal process when the gas is
Easy
(a) Internal energy of an ideal gas is given by
$U = \frac{f}{2}\mu RT = \frac{f}{2}\left( {\frac{N}{{{N_A}}}} \right)\,RT$ ==> $U \propto NT.$
In isothermal process $T =$ constant ==> $U \propto N.$
i.e. internal energy increases by increasing number of molecules $(N).$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 2A certain mass of gas at $273 K$ is expanded to $81$ times its volume under adiabatic condition. If $\gamma = 1.25$ for the gas, then its final temperature is ..... $^oC$View Solution
- 3The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel isView Solution
- 4A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 5$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 6View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
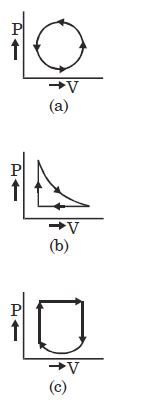
- 7The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
- 8A refrigerator consumes an average $35\, {W}$ power to operate between temperature $-10^{\circ} {C}$ to $25^{\circ} {C}$. If there is no loss of energy then how much average heat per second does it transfer? (in ${J} / {s}$)View Solution
- 9A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
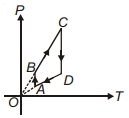
- 10An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
