In a vessel, the gas is at pressure $P_0$. If the mass of all the molecules is halved and their speed is doubled, then the resultant pressure will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of an ideal gas is increased from $27°C$ to $927°C.$ The root mean square speed of its molecules becomesView Solution
- 2View SolutionThe number of translational degrees of freedom for a diatomic gas is
- 3One mole of a monoatomic gas is mixed with three moles of a diatomic gas. The molecular specific heat of mixture at constant volume is $\frac{\alpha^{2}}{4} R J / mol\,K$; then the value of $\alpha$ will be $.......$ (Assume that the given diatomic gas has no vibrational mode.)View Solution
- 4An ideal gas initially at pressure $1$ bar is being compressed from $30 \,m ^{3}$ to $10\, m ^{3}$ volume and its temperature decreases from $320\, K$ to $280\, K$. then find final pressure of gas (in bar)View Solution
- 5When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 6At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
- 7Match List$-I$ with List$-II:$View Solution
List$-I$ List$-II$ $(A)$ $3$ Translational degrees of freedom $(I)$ Monoatomic gases $(B)$ $3$ Translational,$2$ rotational degrees of freedoms $(II)$ Polyatomic gases $(C)$ $3$ Translational,$2$ rotational and $1$ vibrational degrees of freedom $(III)$ Rigid diatomic gases $(D)$ $3$ Translational,$3$ rotational and more than one vibrational degrees of freedom $(IV)$ Nonrigid diatomic gases Choose the correct answer from the options given below:
- 8Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
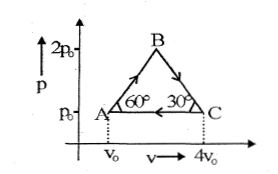
- 9The equation of a certain gas can be written as: ${\left( {\frac{{{T^7}}}{{{P^2}}}} \right)^{1/5}} = $ constant. The specific heat at constant volume of this gas is in $\left( {in\frac{J}{{mol\,K}}} \right)$View Solution
- 10On giving equal amount of heat at constant volume to $1\, mol$ of a monoatomic and a diatomic gas the rise in temperature $(\Delta T)$ is more forView Solution
