Match List$-I$ with List$-II:$
| List$-I$ | List$-II$ |
| $(A)$ $3$ Translational degrees of freedom | $(I)$ Monoatomic gases |
| $(B)$ $3$ Translational,$2$ rotational degrees of freedoms | $(II)$ Polyatomic gases |
| $(C)$ $3$ Translational,$2$ rotational and $1$ vibrational degrees of freedom | $(III)$ Rigid diatomic gases |
| $(D)$ $3$ Translational,$3$ rotational and more than one vibrational degrees of freedom | $(IV)$ Nonrigid diatomic gases |
Choose the correct answer from the options given below:
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):View Solution
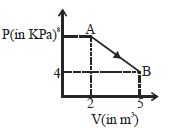
- 2A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 3At $NTP$ the mass of one litre of air is $1.293\, gm.$ The value of specific gas constant will be ...... $J/K-gm$View Solution
- 4A gas is filled in a vessel at a pressure given by $P = \left( {6.02 \times {{10}^{23}}} \right)kT$ where $k$ is the Boltzmann constant and $T$ is the absolute temperature. The number of molecules per unit volume of the gas isView Solution
- 5A hydrogen cylinder is designed to withstand an internal pressure of $100 \,atm$. At $27^{\circ} C$, hydrogen is pumped into the cylinder which exerts a pressure of $20 \,atm$. At what temperature does the danger of explosion first sets in ......... $K$View Solution
- 6A mixture of $2$ moles of helium gas (atomic mass $=4 \ amu$ ), and $1$ mole of argon gas (atomic mass $=40 \ amu$ ) is kept at $300 \ K$ in a container. The ratio of the rms speeds $\left(\frac{\left. v _{ mms } \text { (helium }\right)}{ v _{\text {rms }} \text { (argon) }}\right)$ is :View Solution
- 7If $C_p$ and $C_v$ denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, thenView Solution
- 8The plot that depicts the behavior of the mean free time $t$ (time between two successive collisions) for the molecules of an ideal gas, as a function of temperature $(T)$, qualitatively, is (Graphs are schematic and not drawn to scale)View Solution
- 9At a certain temperature, the $r.m.s.$ velocity for ${O_2}$ is $400\, m/sec.$ At the same temperature, the $r.m.s.$ velocity for ${H_2}$ molecules will be ....... $m/sec$View Solution
- 10$Assertion :$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and its volume.View Solution
$Reason :$ The molecules of a gas collide with each other and the velocities of the molecules change due to the collision.
