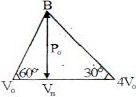
Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature at which the kinetic energy of oxygen molecules becomes double than its value at $27^{\circ}\,C$ is $............^{\circ}\,C$View Solution
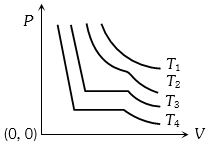
- 2The change in volume $V$ with respect to an increase in pressure $P$ has been shown in the figure for a non-ideal gas at four different temperatures ${T_1},\,{T_2},\,{T_3}$ and ${T_4}$. The critical temperature of the gas isView Solution
- 3View SolutionInternal forces acting in a system of particle can change
- 4A cylinder of capacity $20$ litres is filled with ${H_2}$ gas. The total average kinetic energy of translatory motion of its molecules is $1.5 \times {10^5}\,J$. The pressure of hydrogen in the cylinder isView Solution
- 5A container is divided into two chambers by a partition. The volume of first chamber is $4.5$ litre and second chamber is $5.5$ litre. The first chamber contain $3.0$ moles of gas at pressure $2.0\, atm$ and second chamber contain $4.0$ moles of gas at pressure $3.0\, atm$ .After the partition is removed and the mixture attains equilibrium, then, the common equilibrium pressure existing in the mixture is $x \times 10^{-1} atm$. Value of $x$ is.........View Solution
- 6$Assertion$: The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5\, times$ the product of its pressure and its volume.View Solution
$Reason$ : The molecules of a gas collide with each other and the velocities of the molecules change due to the collision. - 7View SolutionThe gas which obeys Boyle's law for maximum range of temperature is
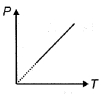
- 8The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]View Solution
- 9We write the relation for Boyle's law in the form $PV = C$ when the temperature remains constant. In this relation, the magnitude of $C$ depends uponView Solution
- 10Consider an ideal gas confined in an isolated closed chamber. As the gas undergoes an adiabatic expansion, the average time of collision between molecules increases as $V ^q$, where $V$ is the volume of the gas. The value of $q$ is $\left( {\gamma = \frac{{{C_P}}}{{{C_V}}}} \right)$View Solution