In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal to
Easy
(d) At constant volume $P \propto T$==> $\frac{{{P_1}}}{{{P_2}}} = \frac{{{T_1}}}{{{T_2}}}$
==>$\frac{{{P_1}}}{{{P_2}}} = \frac{{300}}{{400}} = \frac{3}{4}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
- 2Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 3A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 4The work of $146\ kJ$ is performed in order to compress one kilo mole of gas adiabatically and in this process the temperature of the gas increases by $7^o C$. The gas is $(R=8.3\ J\ mol^{-1} K^{-1})$View Solution
- 5$1\,g$ of a liquid is converted to vapour at $3 \times 10^5\,Pa$ pressure. If $10 \%$ of the heat supplied is used for increasing the volume by $1600\,cm ^3$ during this phase change, then the increase in internal energy in the process will be $............\,J$View Solution
- 6In the $P-V$ diagram, $I$ is the initial state and $F$ is the final state. The gas goes from $I$ to $F$ by $(i)\,IAF,\,\,(ii)\,IBF,\,\,(iii) ICF$. The heat absorbed by the gas isView Solution
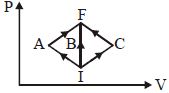
- 7$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 8An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 9The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution
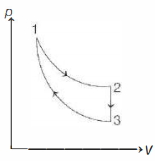
- 10The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
