The work of $146\ kJ$ is performed in order to compress one kilo mole of gas adiabatically and in this process the temperature of the gas increases by $7^o C$. The gas is $(R=8.3\ J\ mol^{-1} K^{-1})$
AIEEE 2006, Medium
$W = \frac{{nR\Delta T}}{{1 - \gamma }} \Rightarrow - 146000 = \frac{{1000 \times 8.3 \times 7}}{{1 - \gamma }}$
$or\,1 - \gamma = - \frac{{58.1}}{{146}} \Rightarrow \gamma = 1 + \frac{{58.1}}{{146}} = 1.4$
Hence the gas is diatomic.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionFor a reversible process, necessary condition is
- 2In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 3Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 4Efficiency of Carnot engine is $100\%$ ifView Solution
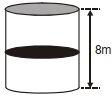
- 5A thermally isolated cylindrical closed vessel of height $8 m$ is kept vertically. It is divided into two equal parts by a diathermic (perfect thermal conductor) frictionless partition of mass $8.3 kg$. Thus the partition is held initially at a distance of $4 m$ from the top, as shown in the schematic figure below. Each of the two parts of the vessel contains $0.1$ mole of an ideal gas at temperature $300 K$. The partition is now released and moves without any gas leaking from one part of the vessel to the other. When equilibrium is reached, the distance of the partition from the top (in $m$ ) will be. . . . . . (take the acceleration due to gravity $=10 ms ^{-2}$ and the universal gas constant $=8.3 J mol ^{-1} K ^{-1}$ ).View Solution
- 6An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
- 7View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
- 8A Carnot’s engine is made to work between $200°C$ and $0°C$ first and then between $0°C$ and $-200°C.$ The ratio of efficiencies of the engine in the two cases isView Solution
- 9During an adiabatic expansion of $2\, moles$ of a gas, the change in internal energy was found $-50J.$ The work done during the process is ...... $J$View Solution
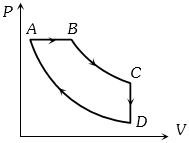
- 10A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution