Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$
JEE MAIN 2023, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
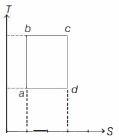
- 1An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
- 2A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 3An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
- 4Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason $R$.View Solution
Assertion $A$ : If $dQ$ and $dW$ represent the heat supplied to the system and the work done on the system respectively. Then according to the first law of thermodynamics $d Q=d U-d W$.
Reason $R :$ First law of thermodynamics is based on law of conservation of energy.
In the light of the above statements, choose the correct answer from the option given below :
- 5A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
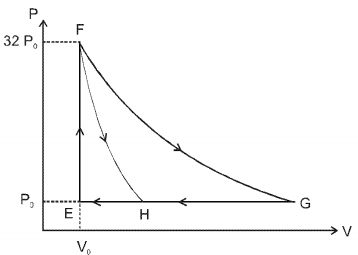
- 6One mole of a monatomic ideal gas is taken along two cyclic processes $E \rightarrow F \rightarrow G \rightarrow E$ and $E \rightarrow F \rightarrow H \rightarrow$ E as shown in the $PV$ diagram. The processes involved are purely isochoric, isobaric, isothermal or adiabatic. $Image$View Solution
Match the paths in List $I$ with the magnitudes of the work done in List $II$ and select the correct answer using the codes given below the lists.
List $I$ List $I$ $P.$ $\quad G \rightarrow E$ $1.$ $\quad 160 P_0 V_0 \ln 2$ $Q.$ $\quad G \rightarrow H$ $2.$ $\quad 36 P _0 V _0$ $R.$ $\quad F \rightarrow H$ $3.$ $\quad 24 P _0 V _0$ $S.$ $\quad F \rightarrow G$ $4.$ $\quad 31 P_0 V_0$ Codes: $ \quad \quad P \quad Q \quad R \quad S $
- 7An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
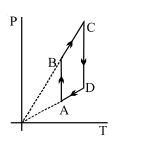
- 8View SolutionPressure versus temperature graph of an ideal gas is shown in figure
- 9$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
- 10An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
