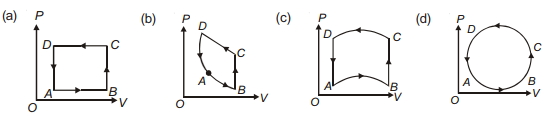
In following figures $(a)$ to $(d)$, variation of volume by change of pressure is shown in figure. The gas is taken along the path $A B C D A$. Change in internal energy of the gas will be .......

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
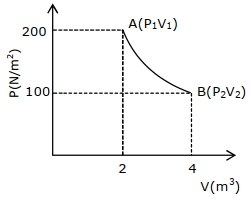
- 1One mole of an ideal gas at $27^{\circ} {C}$ is taken from ${A}$ to ${B}$ as shown in the given ${PV}$ indicator diagram. The work done by the system will be $......\times 10^{-1} \,{J}$View Solution
[Given : $R=8.3\, {J} /\,mole\,{K}, \ln 2=0.6931$ ] (Round off to the nearest integer)
- 2A vertical cylinder with heat-conducting walls is closed at the bottom and is fitted with a smooth light piston. It contains one mole of an ideal gas. The temperature of the gas is always equal to the surrounding’s temperature, $T_0$. The piston is moved up slowly to increase the volume of the gas to $\eta$ times. Which of the following is incorrect?View Solution
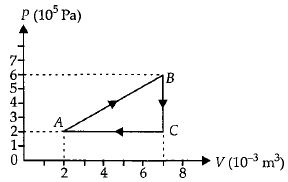
- 3A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 4A mixture of ideal gas containing $5$ moles of monatomic gas and $1$ mole of rigid diatomic gas is initially at pressure $P _0$, volume $V _0$ and temperature $T _0$. If the gas mixture is adiabatically compressed to a volume $V _0 / 4$, then the correct statement(s) is/are,View Solution
(Give $2^{1.2}=2.3 ; 2^{3.2}=9.2 ; R$ is gas constant)
$(1)$ The final pressure of the gas mixture after compression is in between $9 P _0$ and $10 P _0$
$(2)$ The average kinetic energy of the gas mixture after compression is in between $18 RT _0$ and $19 RT _0$
$(3)$ The work $| W |$ done during the process is $13 RT _0$
$(4)$ Adiabatic constant of the gas mixture is $1.6$
- 5View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
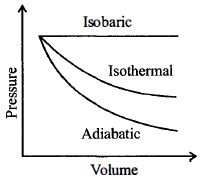
- 6A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
- 7The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution
- 8Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 9Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 10An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
