A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?
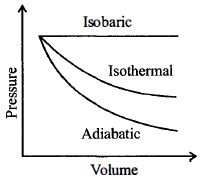

JEE MAIN 2013, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
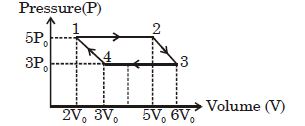
- 2View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
- 3$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 4In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
- 5An ideal gas follows a process described by the equation $PV ^2= C$ from the initial $\left( P _1, V _1, T _1\right)$ to final $\left(P_2, V_2, T_2\right)$ thermodynamics states, where $C$ is a constant. ThenView Solution
- 6Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
- 7A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution
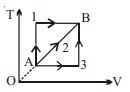
- 8If $300 ml$ of a gas at ${27^o}C$ is cooled to ${7^o}C$ at constant pressure, then its final volume will be ...... $ml$View Solution
- 9In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
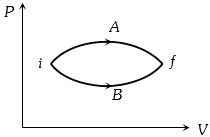
- 10An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
