One mole of an ideal gas at $27^{\circ} {C}$ is taken from ${A}$ to ${B}$ as shown in the given ${PV}$ indicator diagram. The work done by the system will be $......\times 10^{-1} \,{J}$
[Given : $R=8.3\, {J} /\,mole\,{K}, \ln 2=0.6931$ ] (Round off to the nearest integer)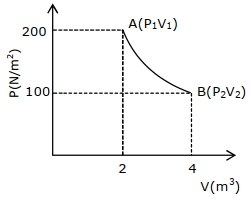
JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A bubble from bottom of lake rises to its surface. Its volume doubles in the process. Assuming isothermal conditions, atmospheric pressure $= 75\, cm$ of $Hg$ and ratio of densities of mercury and water $40/3$. The depth of lake will be ..... $m$View Solution
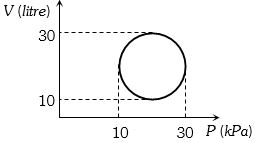
- 2View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
- 3View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
- 4The pressure and density of a diatomic gas $\gamma = 7/5$ change adiabatically from $(P, d)$ to $(P', d').$ If $\frac{{d'}}{d} = 32,$ then $\frac{{P'}}{P}$should beView Solution
- 5An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 6View SolutionBy opening the door of a refrigerator placed inside a room you
- 7$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 8If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
- 9A reversible heat engine converts one-fourth of the heat input into work. When the temperature of the sink is reduced by $52\, K$, its efficiency is doubled. The temperature in Kelvin of the source will be ...... .View Solution
- 10Two identical balls, $A$ and $B$ , of uniform composition and initially at the same temperature, each absorb exactly the same amount of heat. $A$ is hanging down from the ceiling while $B$ rests on the horizontal floor in the same room. Assuming no subsequent heat loss by the balls, which of the following statements is correct about their final temperatures, $T_A$ and $T_B$ , once the balls have reached their final state?View Solution