The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :
JEE MAIN 2024, Diffcult
$\mathrm{dQ}=\mathrm{du}+\mathrm{dW}$
$\mathrm{CdT}=\mathrm{C}_{\mathrm{V}} \mathrm{dT}+\mathrm{PdV}$ $....(1)$
$\therefore \quad \mathrm{PV}^2=\mathrm{RT}$
$\quad \mathrm{P}=\text { constant }$
$\quad \mathrm{P}(2 \mathrm{VdV})=\mathrm{RdT}$
$\quad \mathrm{PdV}=\frac{\mathrm{RdT}}{2 \mathrm{~V}}$
Put in equation $(1)$
$\mathrm{C}=\mathrm{C}_{\mathrm{V}}+\frac{\mathrm{R}}{2 \mathrm{~V}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe isothermal bulk modulus of a perfect gas at normal pressure is
- 2$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
- 3An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 4The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
- 5An engine operating between the boiling and freezing points of water will haveView Solution
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
- 6View SolutionThe process in which no heat enters or leaves the system is termed as
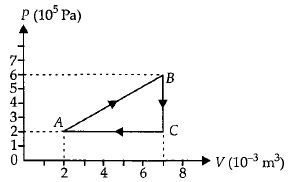
- 7A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 8View SolutionThe specific heat of a gas in an isothermal process is
- 9One mole of an ideal gas with $\gamma = 1.4$, is adiabatically compressed so that its temperature rises from $27°C$ to $35°C$. The change in the internal energy of the gas is ....... $J$ $(R = 8.3\,J/mol.K)$View Solution
- 10An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
