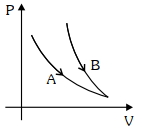
Adiabatic $\Rightarrow \mathrm{PV}^{\prime}=$ const.
Or $\mathrm{P}\left(\frac{\mathrm{T}}{\mathrm{P}}\right)^{\mathrm{r}}=$ const.
$\frac{\mathrm{T}^v}{\mathrm{P}^{v-1}}=\text { const. }$
Curve $(A)$ is isothermal
$\mathrm{T}=$ const.
$\mathrm{PV}=$ const.
Download our appand get started for free
Similar Questions
- 1Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 2For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
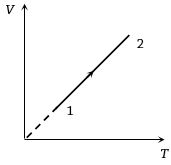
- 3Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 4If $Q , E$ and $W$ denote respectively the heat added, change in internal energy and the work done by a closed cycle process, thenView Solution
- 5An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
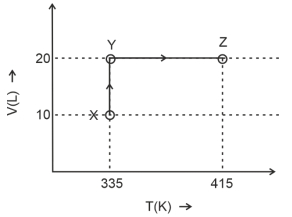
- 6Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
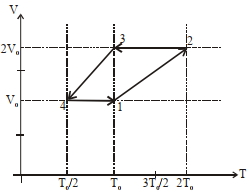
- 7One mole of a monoatomic ideal gas goes through a thermodynamic cycle, as shown in the volume versus temperature ($V-T$) diagram. The correct statement($s$) is/are :View Solution
[ $R$ is the gas constant]
$(1)$ Work done in this thermodynamic cycle $(1 \rightarrow 2 \rightarrow 3 \rightarrow 4 \rightarrow 1)$ is $| W |=\frac{1}{2} RT _0$
$(2)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $2 \rightarrow 3$ is $\left|\frac{ Q _{1 \rightarrow 2}}{ Q _{2 \rightarrow 3}}\right|=\frac{5}{3}$
$(3)$ The above thermodynamic cycle exhibits only isochoric and adiabatic processes.
$(4)$ The ratio of heat transfer during processes $1 \rightarrow 2$ and $3 \rightarrow 4$ is $\left|\frac{Q_{U \rightarrow 2}}{Q_{3 \rightarrow 4}}\right|=\frac{1}{2}$
- 8The work of $146\ kJ$ is performed in order to compress one kilo mole of gas adiabatically and in this process the temperature of the gas increases by $7^o C$. The gas is $(R=8.3\ J\ mol^{-1} K^{-1})$View Solution
- 9View SolutionA measure of the degree of disorder of a system is known as
- 10View SolutionThe rate of recombination or generation are governed by the law(s) of
