In thermodynamics, heat and work are
JEE MAIN 2021, Medium
Heat and work are treated as path functions in thermodynamics.
$\Delta Q =\Delta U +\Delta W$
Since work done by gas depends on type of
process i.e. path and $\Delta U$ depends just on initial
and final states, so $\Delta Q$ i.e. heat, also has to
depend on process is path.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
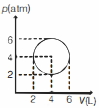
- 2An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
- 3View SolutionWhich is the correct statement
- 4A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
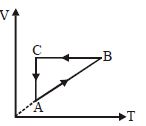
- 5A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
- 6Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
- 7A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 8$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 9$5.6$ liter of helium gas at $STP$ is adiabatically compressed to $0.7$ liter. Taking the initial temperature to be $T _1$, the work done in the process isView Solution
- 10One mole of helium is adiabatically expanded from its initial state $({P_i},{V_i},{T_i})$ to its final state $({P_f},{V_f},{T_f})$. The decrease in the internal energy associated with this expansion is equal toView Solution
