Increase in temperature of a gas filled in a container would lead to
NEET 2019, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A partition divides a container having insulated walls into two compartments $I$ and $II$. the same gas fills the two compartments. The ratio of the number of molecules in compartments $I$ and $II$ isView Solution

- 2A gas is collected over the water at $25°C.$ The total pressure of moist gas was $735 \,mm$ of mercury. If the aqueous vapour pressure at $25°C$ is $23.8\, mm.$ Then the pressure of dry gas is..... $mm$View Solution
- 3When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 4In an ideal gas at temperature $T,$ the average force that a molecule applies on the walls of a closed container depends on $T$ as $T^q$ . A good estimate for $q$ isView Solution
- 5View SolutionMolar specific heat at constant volume, for a non-linear triatomic gas is (vibration mode neglected)
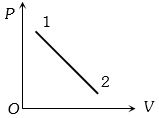
- 6A volume $V$ and pressure $P$ diagram was obtained from state $1$ to state $2$ when a given mass of a gas is subjected to temperature changes. During this process the gas isView Solution
- 7View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
- 8View SolutionWhich law states that effect of pressure is same for all portion
- 9A perfect gas at $27\,^oC$ is heated at constant pressure so as to double its volume. The final temperature of the gas will be, close to ...... $^oC$View Solution
- 10View SolutionAt a given temperature the root mean square velocities of oxygen and hydrogen molecules are in the ratio
