A gas is collected over the water at $25°C.$ The total pressure of moist gas was $735 \,mm$ of mercury. If the aqueous vapour pressure at $25°C$ is $23.8\, mm.$ Then the pressure of dry gas is..... $mm$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The gas in vessel is subjected to a pressure of $20$ atmosphere at a temperature $27°C.$ The pressure of the gas in a vessel after one half of the gas is released from the vessel and the temperature of the remainder is raised by $50°C$ is ....... $atm$View Solution
- 2View SolutionMolecules of a gas behave like
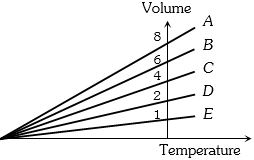
- 3The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
- 4The number density of molecules of a gas depends on their distance $r$ from the origin as, $n\left( r \right) = {n_0}{e^{ - \alpha {r^4}}}$. Then the total number of molecules is proportional toView Solution
- 5Air is filled in a bottle at atmospheric pressure and it is corked at $35°C.$ If the cork can come out at $3$ atmospheric pressure than upto what temperature should the bottle be heated in order to remove the cork ...... $^oC$View Solution
- 6In the kinetic theory of gases, which of these statements is/are true ?View Solution
$(i)$ The pressure of a gas is proportional to the mean speed of the molecules.
$(ii)$ The root mean square speed of the molecules is proportional to the pressure.
$(iii)$ The rate of diffusion is proportional to the mean speed of the molecules.
$(iv)$ The mean translational kinetic energy of a gas is proportional to its kelvin temperature.
- 7A given sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T.$ The mass of each molecule of the gas is $m.$ Which of the following gives the density of the gas $?$View Solution
- 8For an ideal gas the instantaneous change in pressure $'{p} '$ with volume $'v'$ is given by the equation $\frac{{dp}}{{dv}}=-{ap} .$ If ${p}={p}_{0}$ at ${v}=0$ is the given boundary condition, then the maximum temperature one mole of gas can attain is :View Solution
(Here ${R}$ is the gas constant)
- 9In Vander Waal’s equation $a$ and $b$ represent $\left( {P + \frac{a}{{{V^2}}}} \right)\,(V - b) = RT$View Solution
- 10View SolutionFor an ideal gas of diatomic molecules
