Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in addition
KVPY 2011, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In an isolated system the entropy increases.View Solution
$Reason :$ The processes in an isolated system are adiabatic. - 2An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 3In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 J$ of heat and $8J$ of work is done on the gas. If the initial internal energy of the gas was $30J.$ The final internal energy will be ...... $J$View Solution
- 4If $R =$ universal gas constant, the amount of heat needed to raise the temperature of $2$ mole of an ideal monoatomic gas from $273K$ to $373K$ when no work is done ...... $R$View Solution
- 5A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$View Solution
- 6A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 7The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 8The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
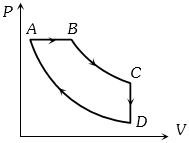
- 9A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 10One mole of an ideal gas at initial temperature $T$, undergoes a quasi-static process during which the volume $V$ is doubled. During the process, the internal energy $U$ obeys the equation $U=a V^3$, where $a$ is a constant. The work done during this process isView Solution
