Match List $I$ with List $II$ :
| List $I$ | List $II$ |
| $A$ Isothermal Process | $I$ Work done by the gas decreases internal energy |
| $B$ Adiabatic Process | $II$ No change in internal energy |
| $C$ Isochoric Process | $III$ The heat absorbed goes partly to increase internal energy and partly to do work |
| $D$ Isobaric Process | $IV$ No work is done on or by the gas |
Choose the correct answer from the options given below :
JEE MAIN 2023, Medium
$\Delta U = nC _{ v } \Delta T$
For isothermal process $T$ is constant
So $\Delta U =0$
$A \longrightarrow II$
Adiabatic process
$\Delta Q =0$
$\Delta Q =\Delta U +\Delta W$
$\quad \Delta U =-\Delta W$
Work done by gas is positive
So $\Delta U$ is negative
$B \longrightarrow I$
For Isochoric process $\Delta W =0$
$C \longrightarrow IV$
For Isobaric process
$\Delta W = P \Delta V \neq 0$
$\Delta U = nC _{ V } \Delta T \neq 0$
Heat absorbed goes partly to increase internal energy and partly do work.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 2$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
- 3View SolutionFirst law of thermnodynamics is given by
- 4View SolutionWhich of the following is not a thermodynamics co-ordinate
- 5View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
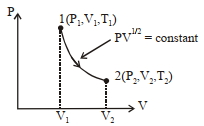
- 6Thermodynamic process is shown below on a $P-V$ diagram for one mole of an ideal gas. If $V _{2}=2 V _{1}$ then the ratio of temperature $T _{2} / T _{1}$ is ...... .View Solution
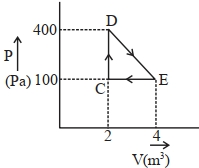
- 7A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
- 8A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 9In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 10When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
