In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy is
Easy
(b) $\Delta Q = \Delta U + \Delta W$
==> $\Delta U = \Delta Q - \Delta W = Q - W$ (using proper sign)
==> $\Delta U = \Delta Q - \Delta W = Q - W$ (using proper sign)
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 2View SolutionThe work done in which of the following processes is equal to the internal energy of the system?
- 3If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
- 4An ideal gas goes from state $A$ to state $B$ via three different processes as indicated in the $P-V$ diagram. If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$View Solution
indicate the change in internal energy along the three processes respectively, then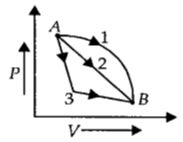
- 5$300 \,cal$. of heat is given to a heat engine and it rejects $225 \,cal$. of heat. If source temperature is $227^{\circ} C$, then the temperature of sink will be____${ }^{\circ} C$.View Solution
- 6An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
- 7An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)View Solution
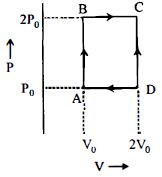
- 8A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 9A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 10View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
